
calculate the number of unpaired electrons in Ti3+ , Mn2+ and calculate the spin only magnetic - Brainly.in

In the fallowing reaction: Cr2O7^2 - (aq) + SO3^2 - (aq) + 8H^+→ 2Cr^3 + + SO4^2 - + H2O the stoichiometric coefficient of SO3^2 - is:
![The spin only magnetic moment value of `[MnBr_(4)]^(2-)` ion is `5.9BM` On the basis of `VBT` Pr... - YouTube The spin only magnetic moment value of `[MnBr_(4)]^(2-)` ion is `5.9BM` On the basis of `VBT` Pr... - YouTube](https://i.ytimg.com/vi/E3pLlLoxf48/maxresdefault.jpg)
The spin only magnetic moment value of `[MnBr_(4)]^(2-)` ion is `5.9BM` On the basis of `VBT` Pr... - YouTube

Mn2+–Mn2+ Magnetic Coupling Effect on Photoluminescence Revealed by Photomagnetism in CsMnCl3 | The Journal of Physical Chemistry Letters
Calculate the 'spin only' magnetic moment of M2+ (aq) ion (Z = 27). - Sarthaks eConnect | Largest Online Education Community
Calculate the 'spin only' magnetic moment of M2+ (aq) ion (Z = 27). - Sarthaks eConnect | Largest Online Education Community

Magnetic moment of `Cr^(+2)(Z=24),Mn^(+2)(Z=25)` and `Fe^(2+) (Z=26)` are x,y,z. they are in order - YouTube

If hund rule is followed magnetic moment of Fe+2 Mn+ and Cr having 24 electron will be in order - Brainly.in

The correct order of spin only magnetic moment (inBM) for Mn^{2+} Cr^{2+} and Ti^{2+} ions is(a} Mn^{2+}>Ti^{2+}>Cr^{2+} (b) Ti^{2+}>Cr^{2+}>Mn^{2+}(c) Mn^{2+}>Cr^{2+}>T^{2+} (d} Cr^{2+}>Ti^{2+}>Mn^{2+} | Snapsolve
![SOLVED:Calculate the spin-only magnetic moment (Us for the following metal ion. [Note that on the exam you will have to show your work]: High spin Mn2+ SOLVED:Calculate the spin-only magnetic moment (Us for the following metal ion. [Note that on the exam you will have to show your work]: High spin Mn2+](https://cdn.numerade.com/ask_images/bfe7cff006004e7aa567a2395d418232.jpg)

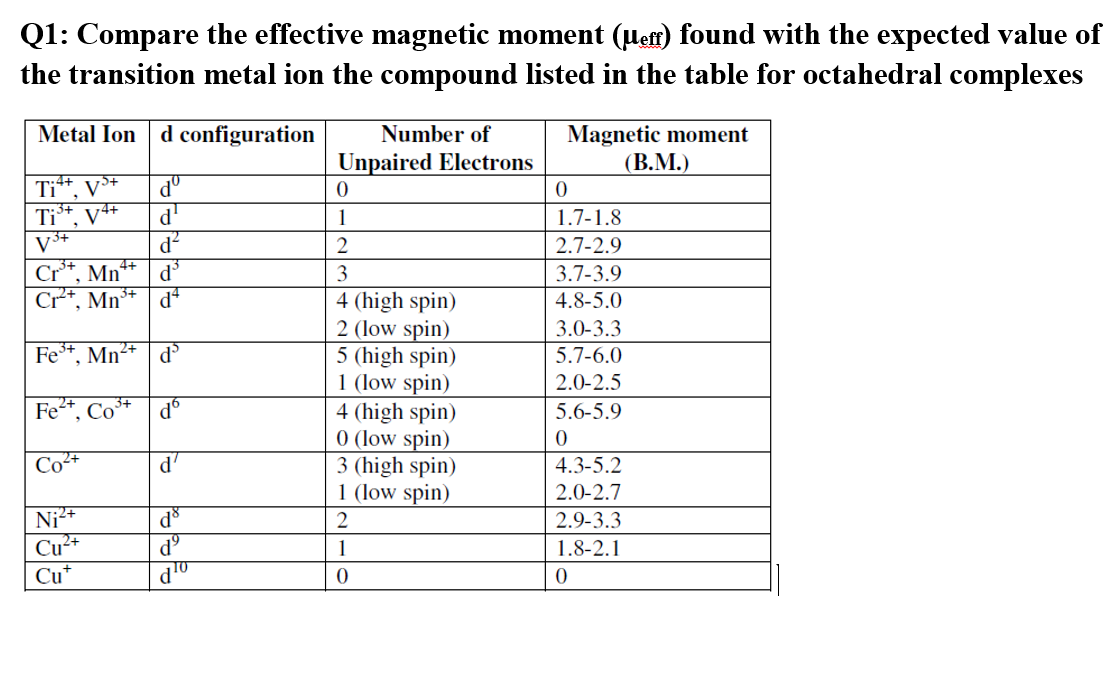

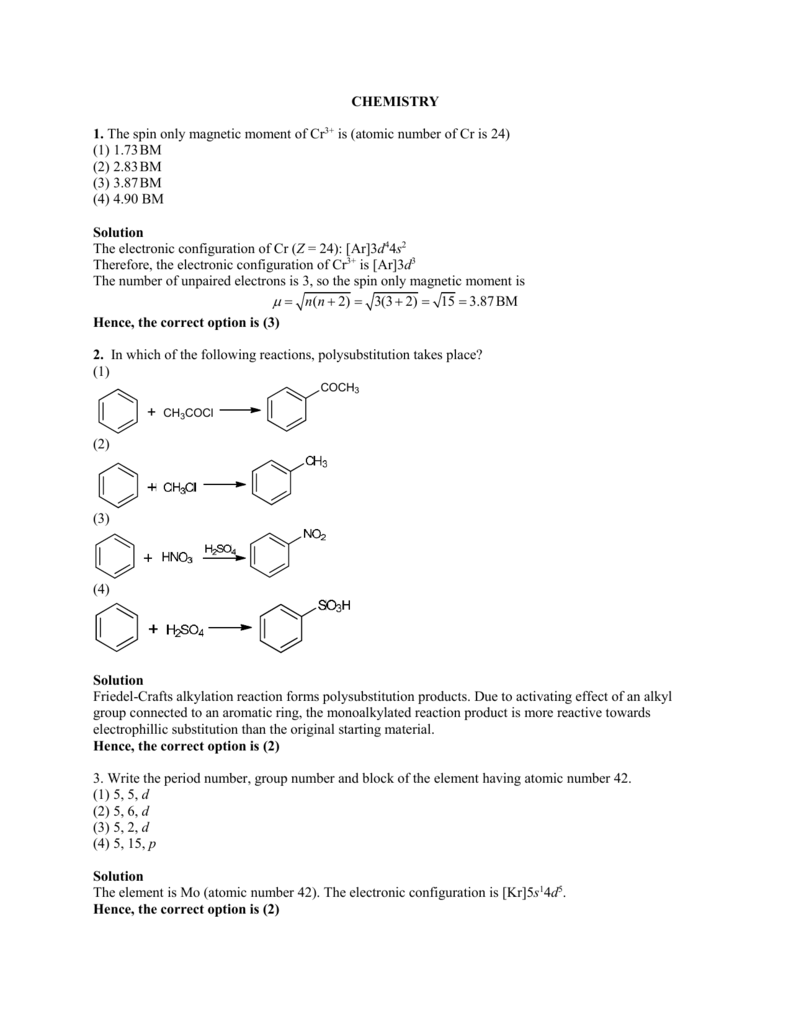








![Expert Answer] Calculate the spin only magnetic moment of mn2+ ion - Brainly.in Expert Answer] Calculate the spin only magnetic moment of mn2+ ion - Brainly.in](https://hi-static.z-dn.net/files/dc8/652c5b211abb681a4d61fb6a49aa9ae7.png)